
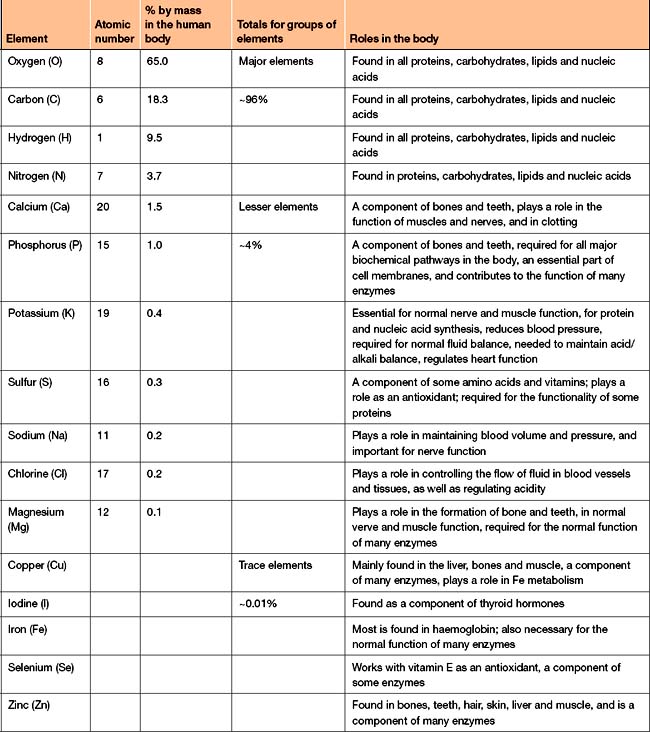
The remaining 4% is composed primarily of calcium, phosphorus, potassium, sulfur, sodium, chlorine, and magnesium, in order of relative abundance. More precisely, 96% of the human body is made up of these four elements. Major Elements and Trace Elements Make up the Human BodyĪll life on Earth contains the elements oxygen, carbon, hydrogen, and nitrogen. By contrast, elements in the same period (i.e., row) are more similar in size and have their electrons located in a similar place, but vary greatly in their chemical properties. Elements in the same group (i.e., column) vary in size but have many chemical properties in common with one another. Elements are organized into columns (groups) and rows (periods) based on other physical and chemical properties, such as reactivity, the location of their outermost electrons, and the ability to make certain types of bonds. The number of protons provides information about the size of an element, but it is not the only organizational principle underlying the structure of the periodic table.
#4 elements that make up 96 of living matter full
The atomic number of an element corresponds to the number of protons found in its nucleus, and each square in the periodic table also provides the full name, chemical symbol, and atomic weight of an element. The periodic table organizes elements based on their physical and chemical properties. The Periodic Table Provides Information about the Physical and Chemical Properties of Elements

Other elements are also necessary for life but only in trace amounts. Living matter is composed primarily of carbon, nitrogen, hydrogen, and oxygen, with smaller amounts of other elements like calcium, phosphorus, potassium, and sulfur. There are 118 known elements, but not all of these are naturally-occurring, and fewer still are essential for life. Elements are the smallest units of matter that cannot be broken down further by chemical processes.
